ELECTRONIC SPECTROSCOPY
IR cannot excite an electronic state. IR photons are so low in energy to excite. Visible can excite to first excited level. UV can excite to second excited electronic state.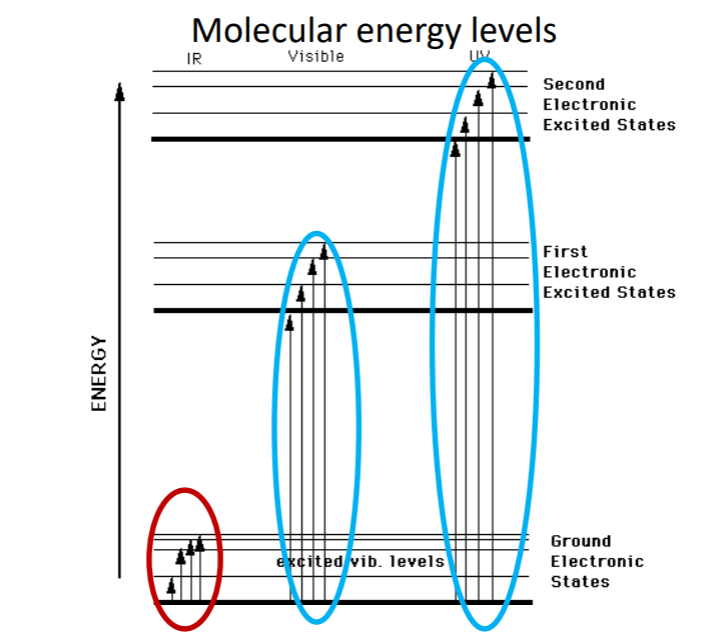 How we investigate electronic transitions?
How we investigate electronic transitions?
UV-Vis absorption spectra
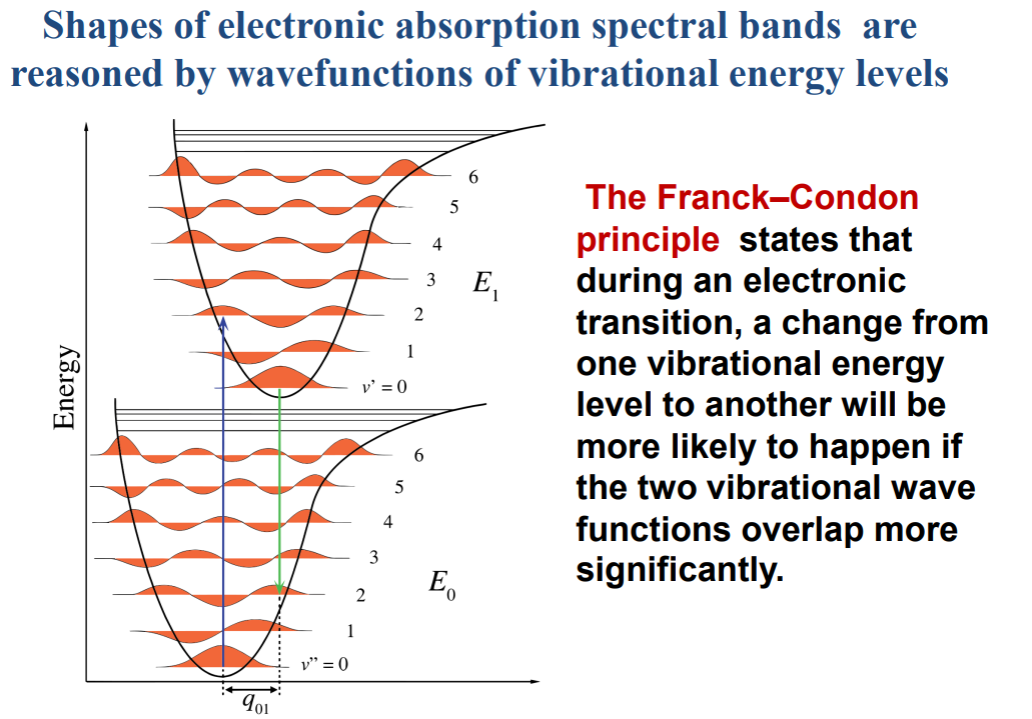
The spectral bands are much broader than the IR absorption or Raman. WHY? Recall what the broader spectral bands refer to. Do not mix it up with the fact of what broader spectral lines refer to! Broad spectral line widths refer to shorter lifetime sof vibrational excitations. Broader spectral band widths refer to many overlapping transitions, a collection of many closely spaced transitions, and that transition involves many final states.Now we can discuss why UV-Vis absorption spectral bands are broader. During an electronic transition, we simultenously access many vibrational levels since electronic transitions are fast (does it related to high uncertainy in where the transition will end at?). Excitations landing in many vibrational states is the Franck-Cordon principle actually.
IMPORTANT!!! Electronic states have shorter lifetimes than vibrational states. WHY???
 Keep questioning this.
Keep questioning this.
Study electron configuration as well.