Further on Raman scattering of polyatomic molecules and implementations of Raman spectroscopy
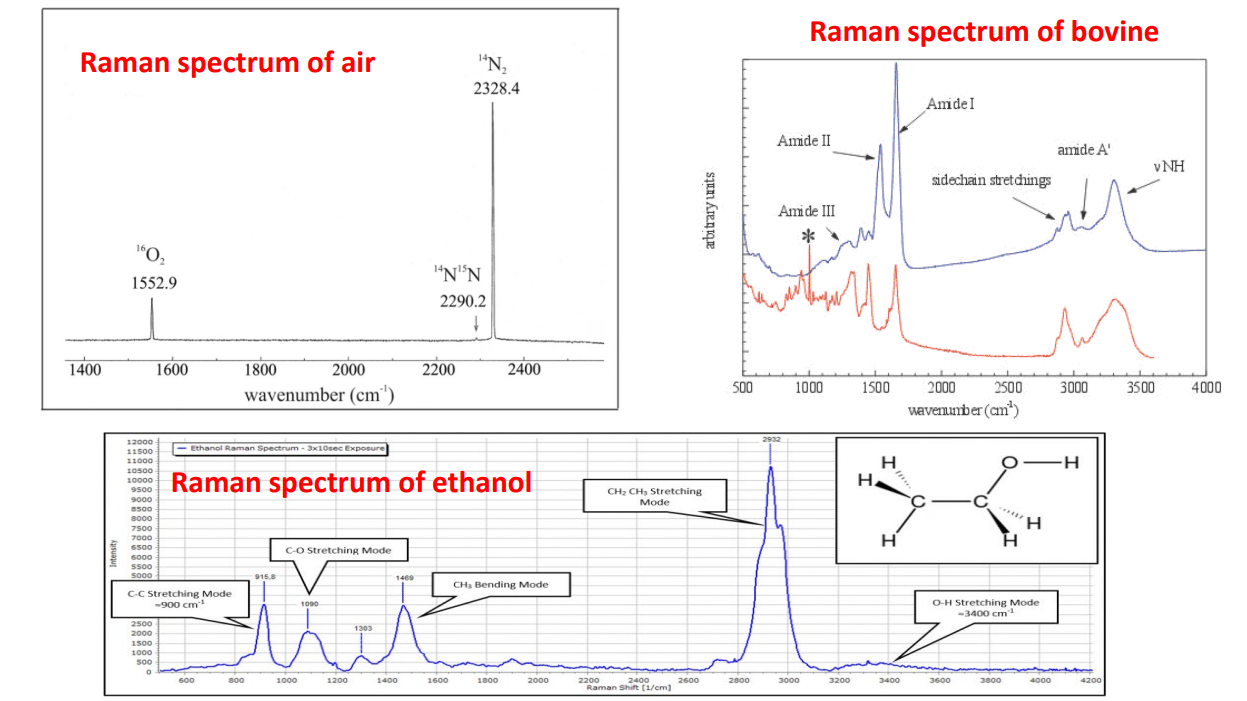 Here we see raman spectrums of air and ethanol (nvm the cow). What you see is stokes shifts for different components.
Here we see raman spectrums of air and ethanol (nvm the cow). What you see is stokes shifts for different components.
Measurable parameters in raman spectral bands are wavenumber, intensity, width, depolarization factor.
What is depolarization factor?
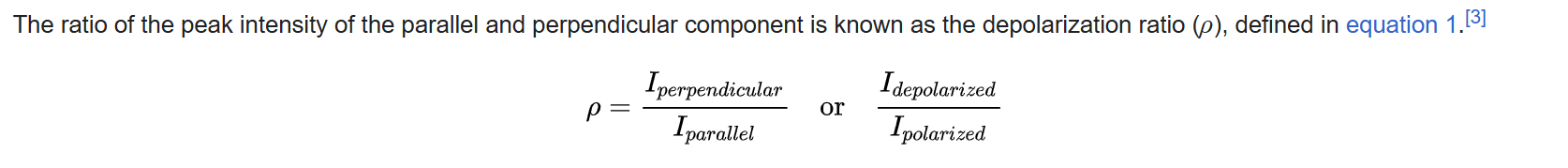
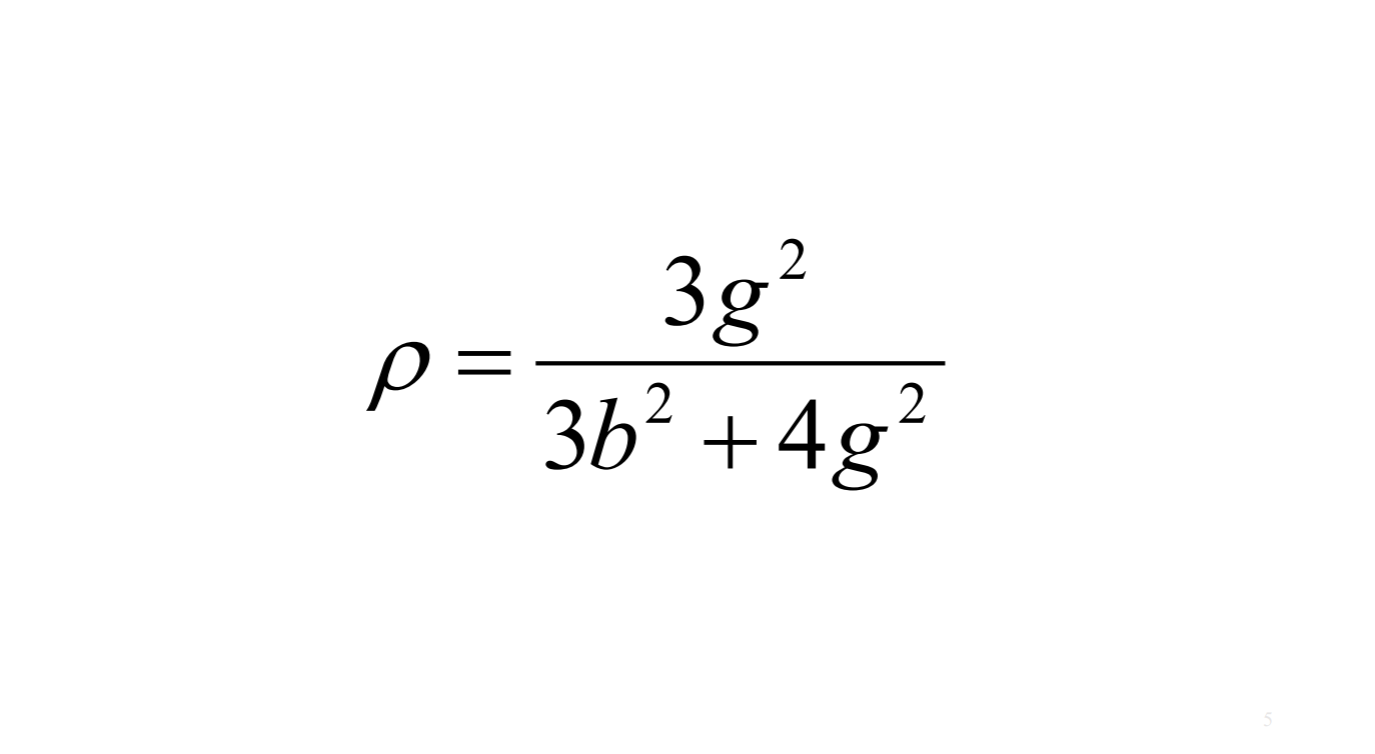 Here, we have Iinpolarizationoutpolarization illustration, and for isotropic samples we have IVH=IHV=IHH
where VH is vertical polarized laser and horizontal polarized Raman; and vice versa.
Here, we have Iinpolarizationoutpolarization illustration, and for isotropic samples we have IVH=IHV=IHH
where VH is vertical polarized laser and horizontal polarized Raman; and vice versa.
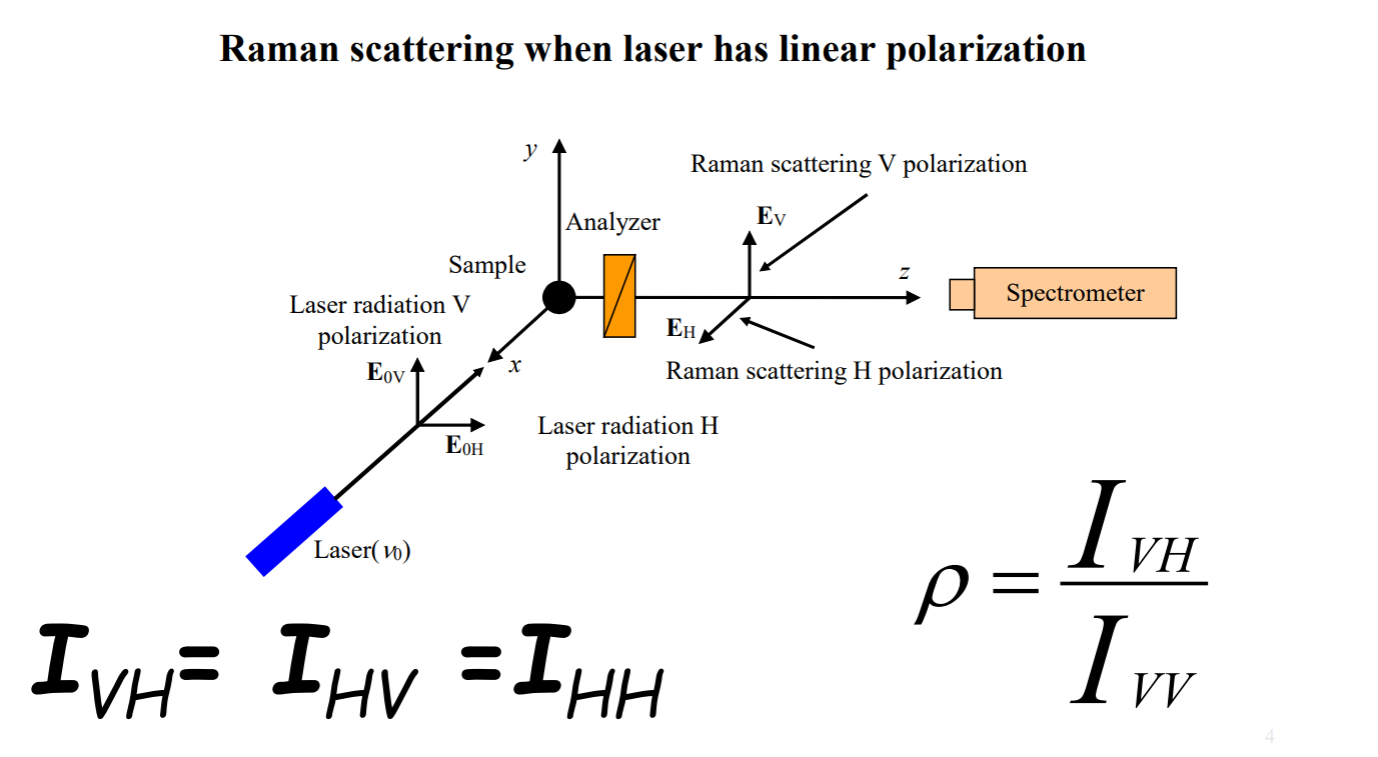
And then we defined or depolarization factor as IVH / IVV which tells us about how much Raman light loses the polarization of incident laser. Then we defined b and g as above, and final defined ρ.
Now looking at the limits, we have:
Totally symmetric modes

Non-totally symmetric modes
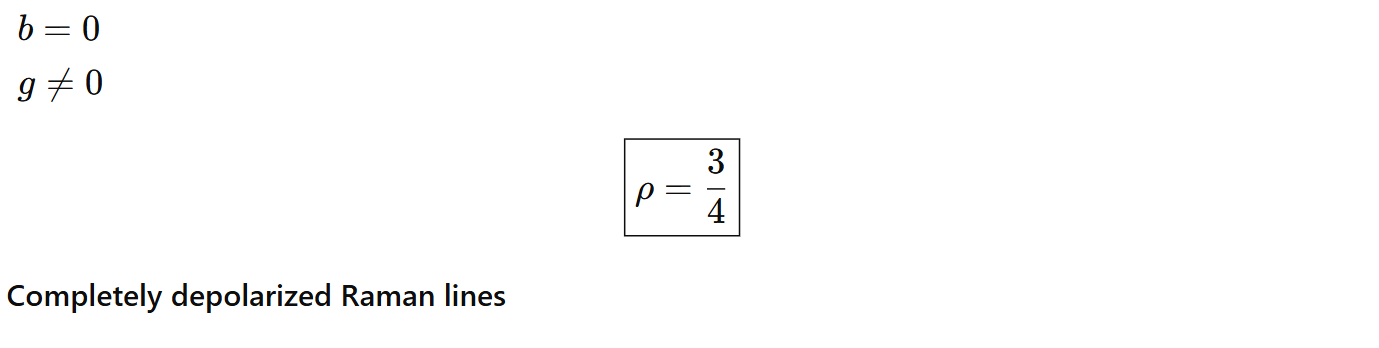 We conclude ρ ≤ 0.75.
Since we can measure IVH and IVV, we can talk about symmetry via Placzek rules.
We conclude ρ ≤ 0.75.
Since we can measure IVH and IVV, we can talk about symmetry via Placzek rules.
Recall: b: isotropic part, g: anisotropic part
Placzek rules
In short, ρ ~ 0 → strongly polarized Raman line, ρ = 0.75 → completely depolarized Raman line
implementations of Raman spectroscopy
There are types of Raman spectroscopy.Conventional (spontaneous) Raman scattering
Resonance Raman scattering
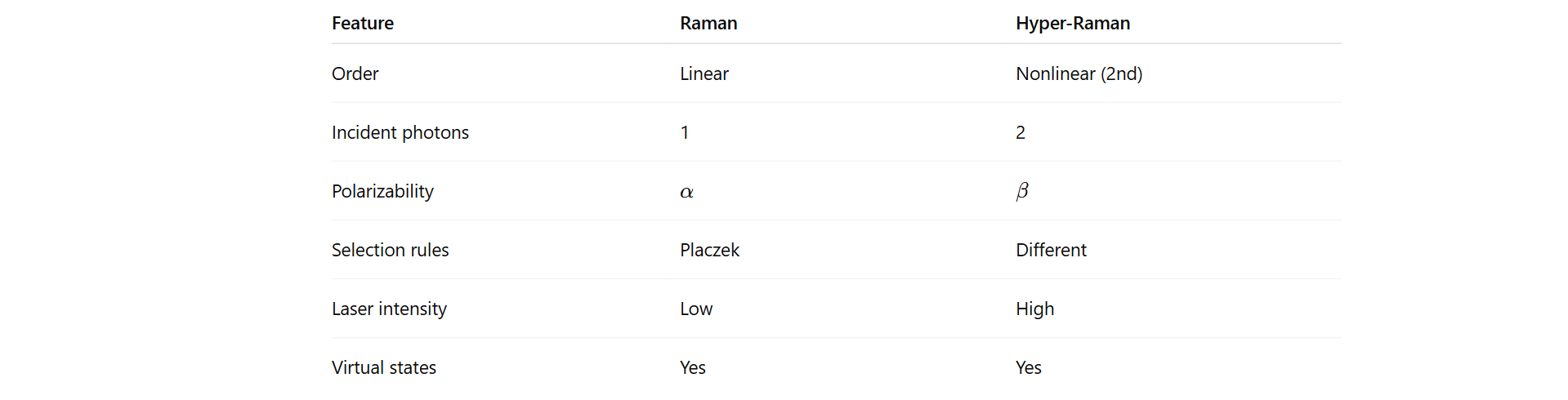
Nonlinear Raman scattering
Let's go.Resonance Raman spectroscopy
Incident laser frequency is very close to electronic excitation energy. The virtual state is nearly an electronic state. Strong vibronic coupling occurs. Resonance raman usually include fluoresence (transitions from real electronic states occurance) background. The closeness in energy leads to greatly increased insensity of Raman scattering.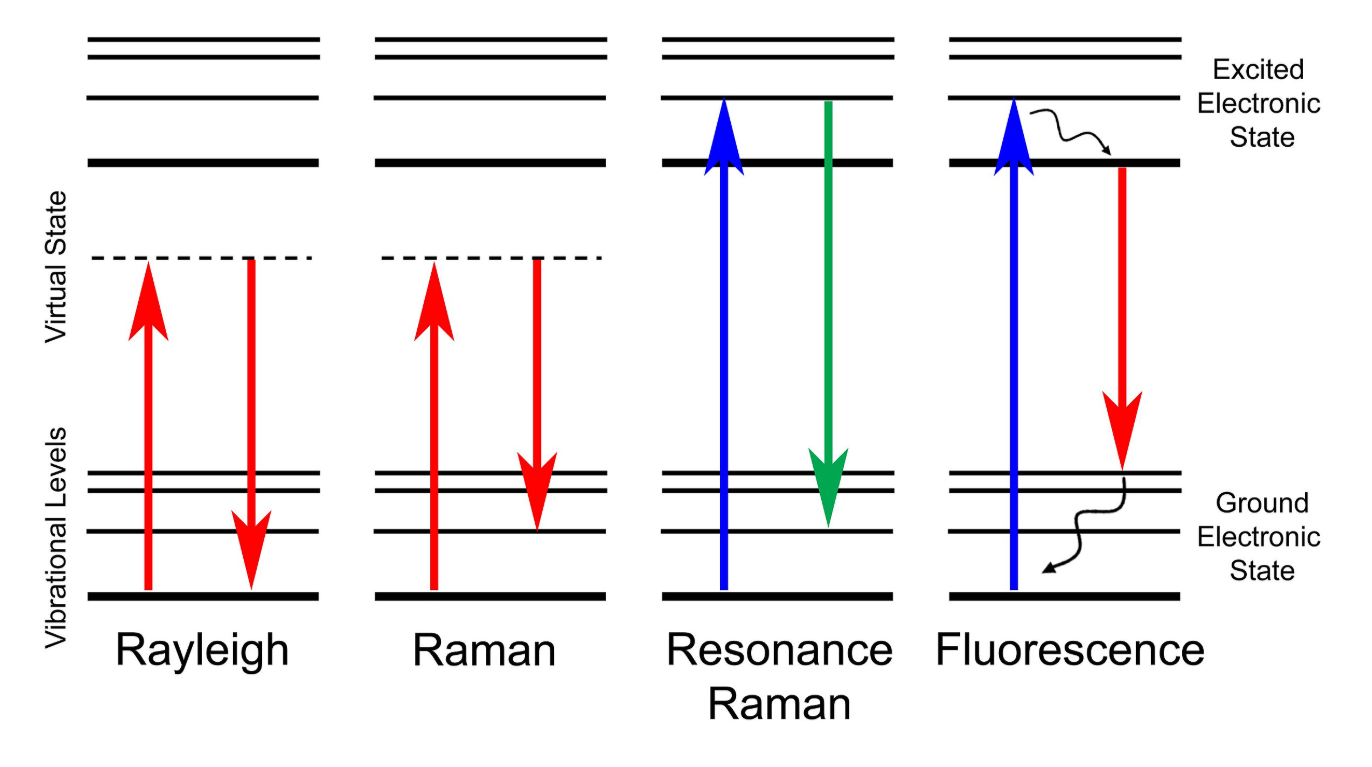
Nonlinear Raman spectroscopy
 Hyper-rayleigh absorbs two photon and emits 2 photon. Same energy. Higher virtual levels compared to normal raman.
Hyper-rayleigh absorbs two photon and emits 2 photon. Same energy. Higher virtual levels compared to normal raman.
On the other hand, in Hyper-raman, two photons are absorbed, molecule reaches a virtual state, one photon is emitted with less energy, the missing energy excites a vibrational mode and shift occurs.

Surface enhanced raman scattering (SERS)
explain... Also mention that HYPERION 3000 and what you can do with it.Chemical imaging using ATR FTIR technique
Used when the preperation of a thin film of the sample is not possible.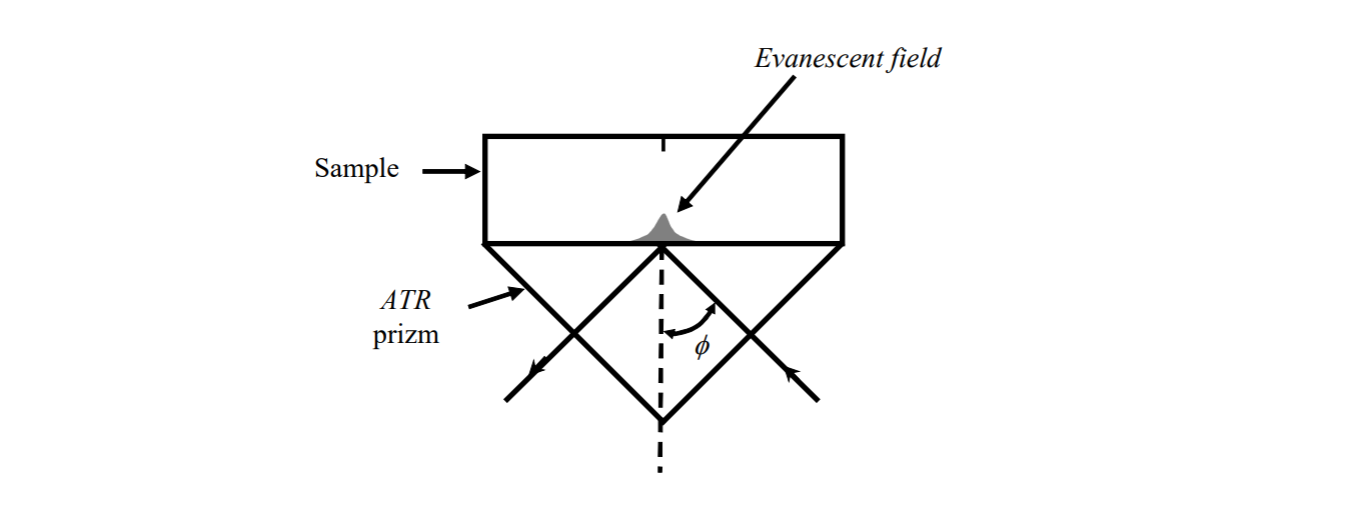 ATR!!!!
ATR!!!!