Raman Spectroscopy
In summary,Vibrational Spectroscopy = Infrared Absorption Spectroscopy + Raman Spectroscopy
Now we'll investigate Raman.Raman Scattering
Here's the Raman experiment.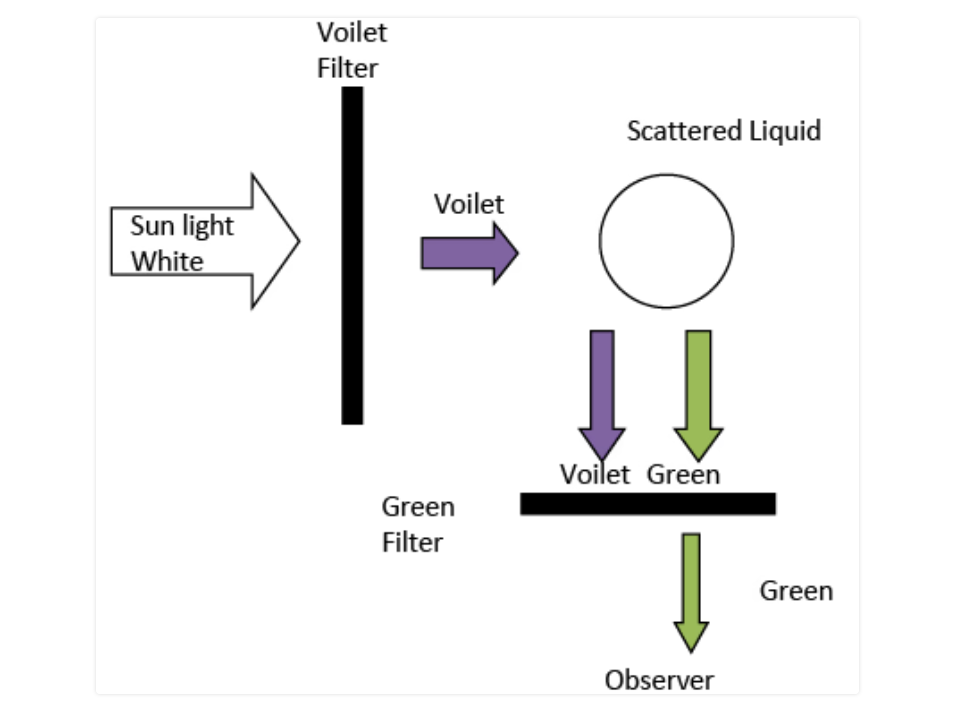 Raman scattering is a quantum mechanical effect by classically illustrated as well. Raman scattering is the inelastic light-matter scattering process, where a photon exchanges energy
with a molecule or a crystal, causing photon's frequency to shift by the energy of a vibrational excitation.
Raman scattering is a quantum mechanical effect by classically illustrated as well. Raman scattering is the inelastic light-matter scattering process, where a photon exchanges energy
with a molecule or a crystal, causing photon's frequency to shift by the energy of a vibrational excitation.
Incoming photon has energy
ℏw0, as it interacts with a molecule it drivens the molecule state to a virtual state, so molecule is temporarily polarized. A virtual state is a state that solution is not included in the SE solved using U(x) of the molecule.
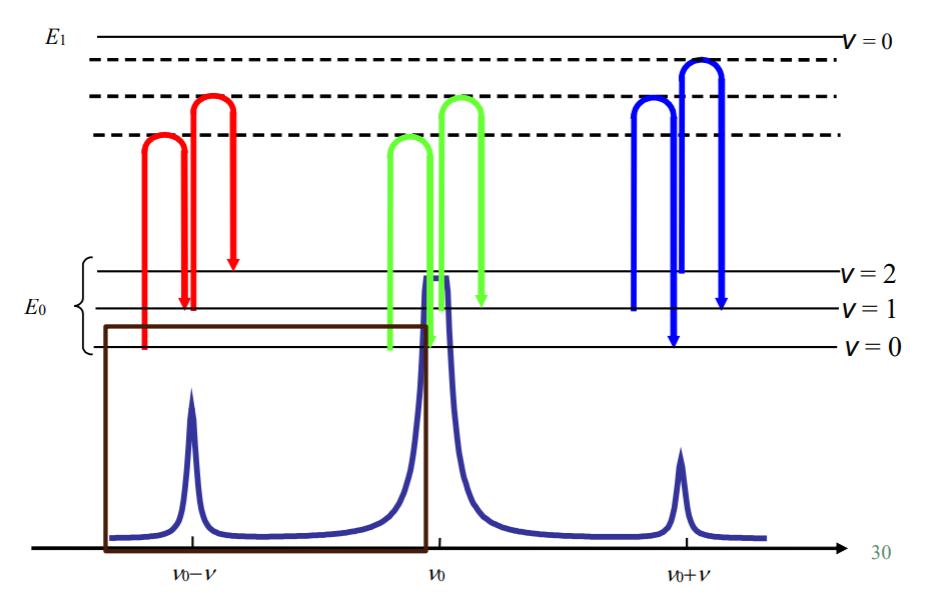
The system reemits the photon with a different energy (a shift),
If it is lower energy, red shift, corresponds to STOKES RAMAN
If it is higher energy, blue shift, corresponds to ANTI-STOKES RAMAN
The energy difference corresponds to vibrational mode of the system:
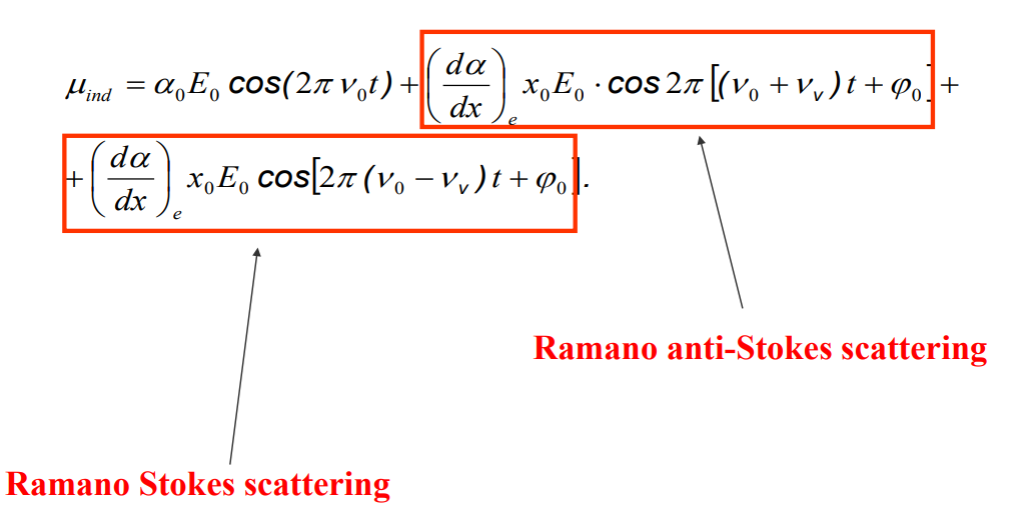
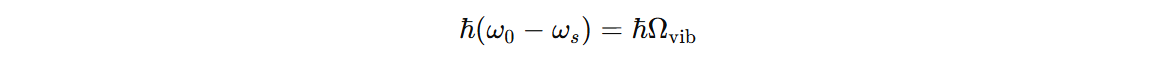 Here we have dipole moment of the molecule, as it interacts with a photon with E field strength. α is polarizibility.
Here we have dipole moment of the molecule, as it interacts with a photon with E field strength. α is polarizibility.
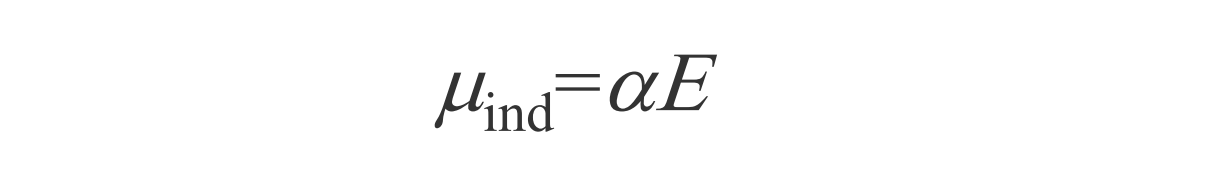
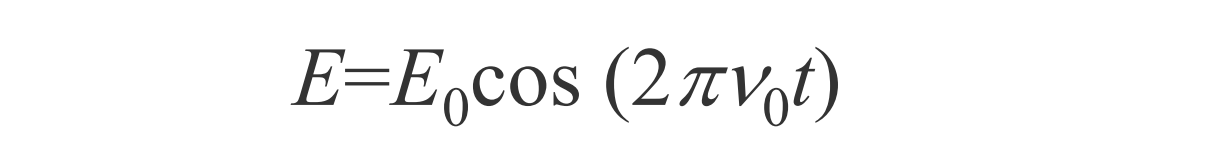 As molecule interacts with photon, dipole moment becomes time dependent. Time dependent dipole, an oscillating dipole, radiates light. That radiation is the scattered light. It results from the interaction of the polarizibility of the molecule and incoming oscillating electric field.
As molecule interacts with photon, dipole moment becomes time dependent. Time dependent dipole, an oscillating dipole, radiates light. That radiation is the scattered light. It results from the interaction of the polarizibility of the molecule and incoming oscillating electric field.
 Raman spectrum of an apple:
Raman spectrum of an apple:
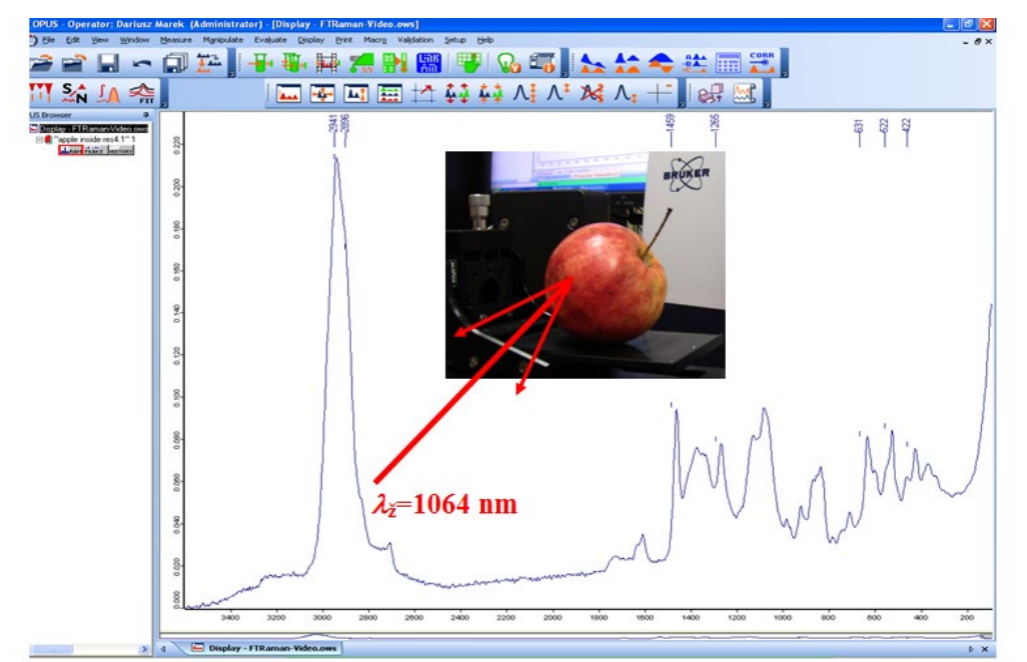
In our spectrum we have the highest peak as the Rayleigh wavelength. Other peaks are the relative shifts. Remember, scattering physically absorption or emission from virtual states.
 In scattering however:
In scattering however:

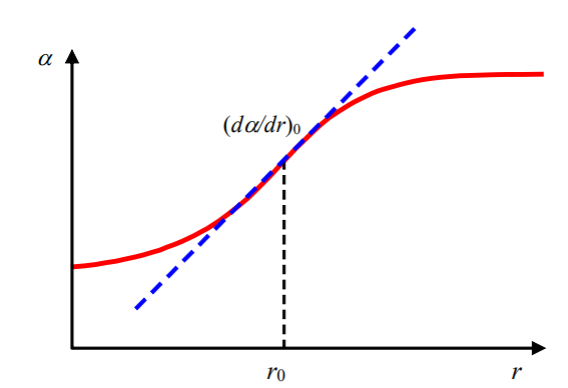
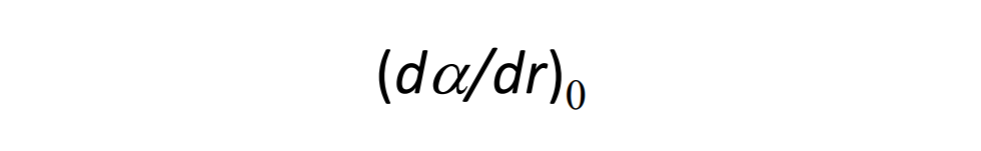 The intensity of Raman scattering of a diatomic molecule depends on change in polarizibility respect to distance. Meali: How strongly the molecule scatters Raman light
depends on how much its polarizibility changes as the bond length or distance from equilibrum changes.
The intensity of Raman scattering of a diatomic molecule depends on change in polarizibility respect to distance. Meali: How strongly the molecule scatters Raman light
depends on how much its polarizibility changes as the bond length or distance from equilibrum changes.
r = internuclear seperation
 Why Rayleigh is the highest peak in Raman spectra?
Why Rayleigh is the highest peak in Raman spectra?
Good question.
Rayleigh is the highest peak because intensity is about probability, not about gaining energy — and anti-Stokes processes are much less probable.
I do remember peaks in Raman corresponded to probabilities. Let's investigate.