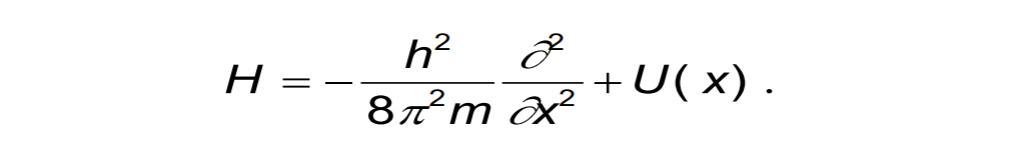Optical Spectroscopy
Introduction to Spectroscopy. We will understand energy levels and transitions between them. Jablonski diagram etc.What is Spectroscopy?
It is a method used by intracting electromagnetic radiation (light etc., when it's light we call it optical spect.) with matter (or substance). It is widely used in materials science for characterization. Mainly for characterization, but also for mechanics, dynamics, kinetics, defects, electronics structure etc.But these are also characteristics, so.
Optical Spectroscopy
We use light to interact with substances. Light in physics are IR, Visible, UV regions.Electromagnetic Radiation
What is electromagnetic radiation? Electromagnetic radiation is the propogating form of coupled magnetic and electric fields. There are no non-propogating coupled EM waves anyways. There are electrostatic and magnetostatic field however. And the coupling terms come from Maxwell's equations, specifically Faraday's Law and Ampere-Maxwell's law: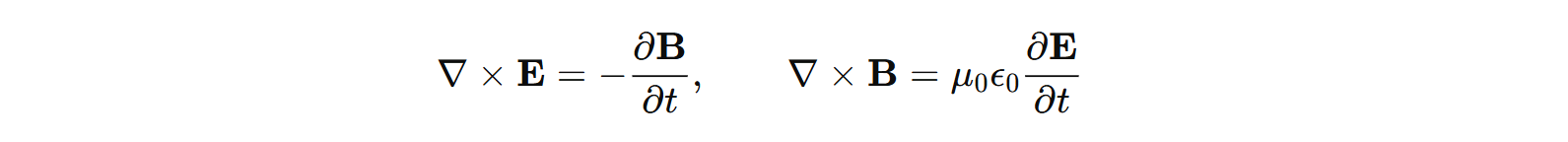 All radiation should be a solution to Maxwell's equations. Not all solutions of Maxwell's equations. Physically, if the solutions obey 1/r behavior,
basically if the fields carry energy away from the source.
All radiation should be a solution to Maxwell's equations. Not all solutions of Maxwell's equations. Physically, if the solutions obey 1/r behavior,
basically if the fields carry energy away from the source.
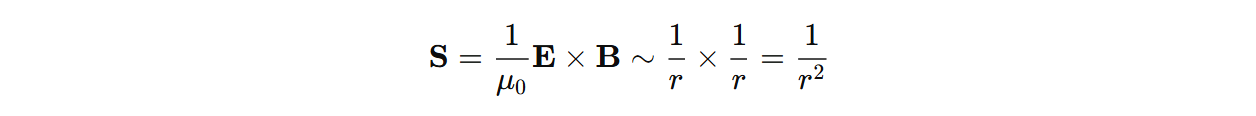 Static fields (E=0, B!=0 or E!=0, B=0) has S=0 and near fields have a time-averaged=0 Poynting vector.
Static fields (E=0, B!=0 or E!=0, B=0) has S=0 and near fields have a time-averaged=0 Poynting vector.
Review Maxwell's equations as well:
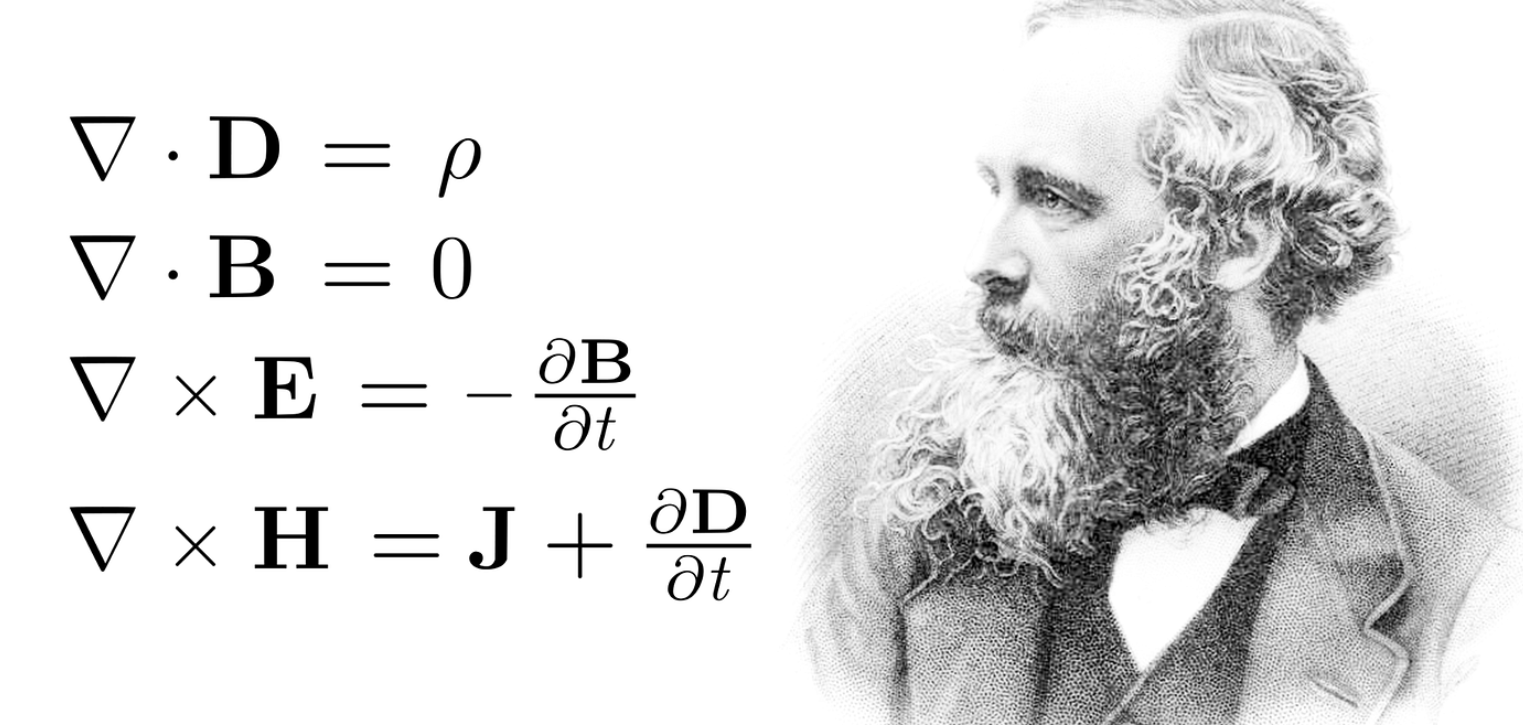 Good review.
Good review.
Understanding spectroscopy is based on Planck equation:
δE = hf
f = v/λ
which comes from the wave equation as a solution to Maxwell's equations: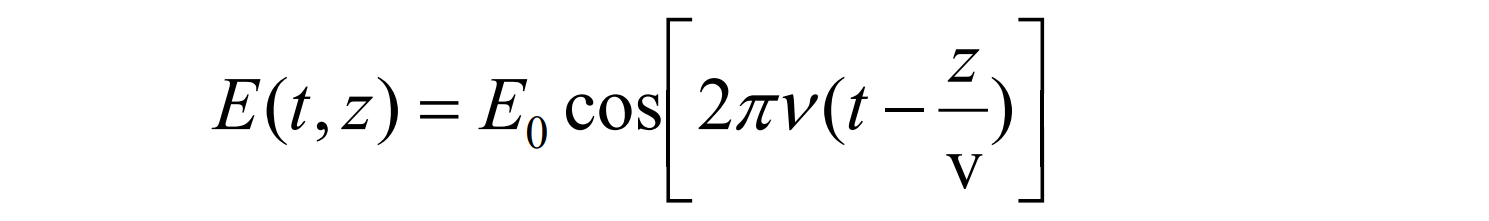 IMPORTANT!!! In some spectroscopies, wavenumber is used instead of wavelength.
IMPORTANT!!! In some spectroscopies, wavenumber is used instead of wavelength.
[wavenumber] = cm-1
Subject of Molecular Spectroscopy
The following represents the types of interaction between Matter and EM Radiation.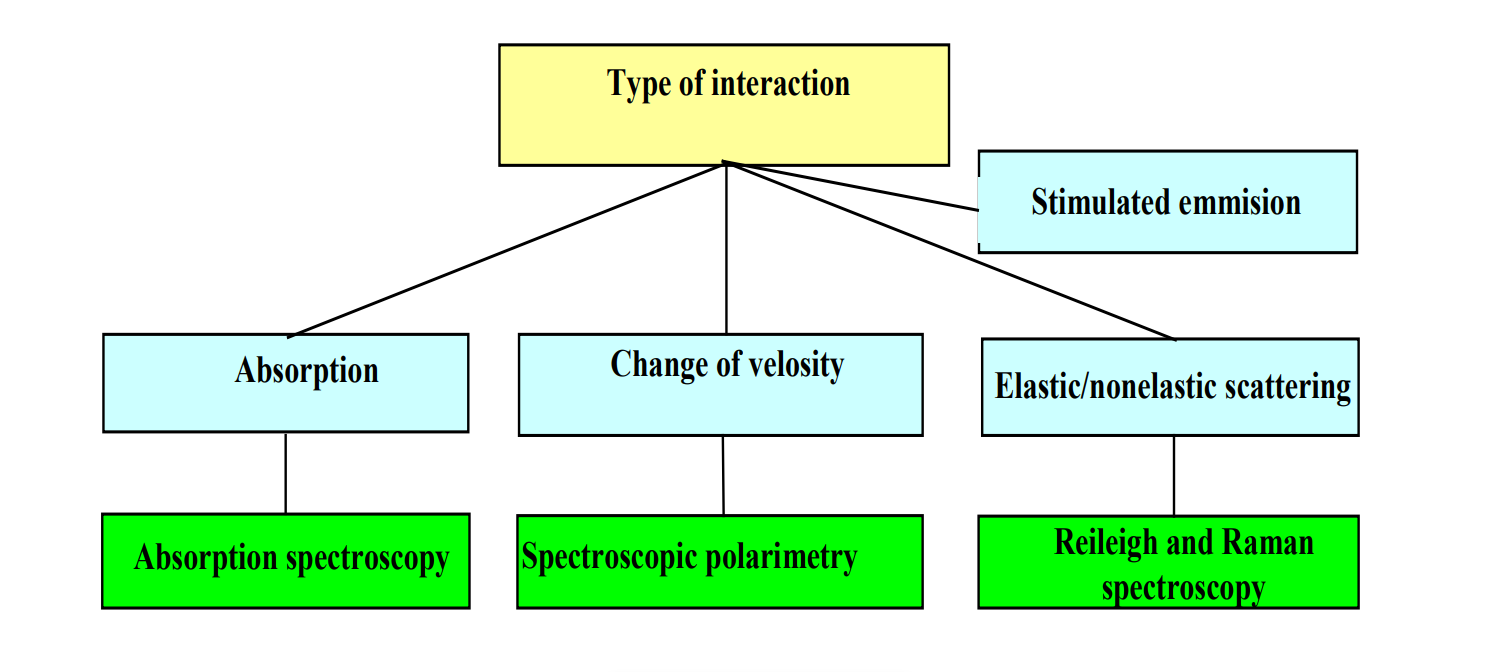
Molecular Energy Levels
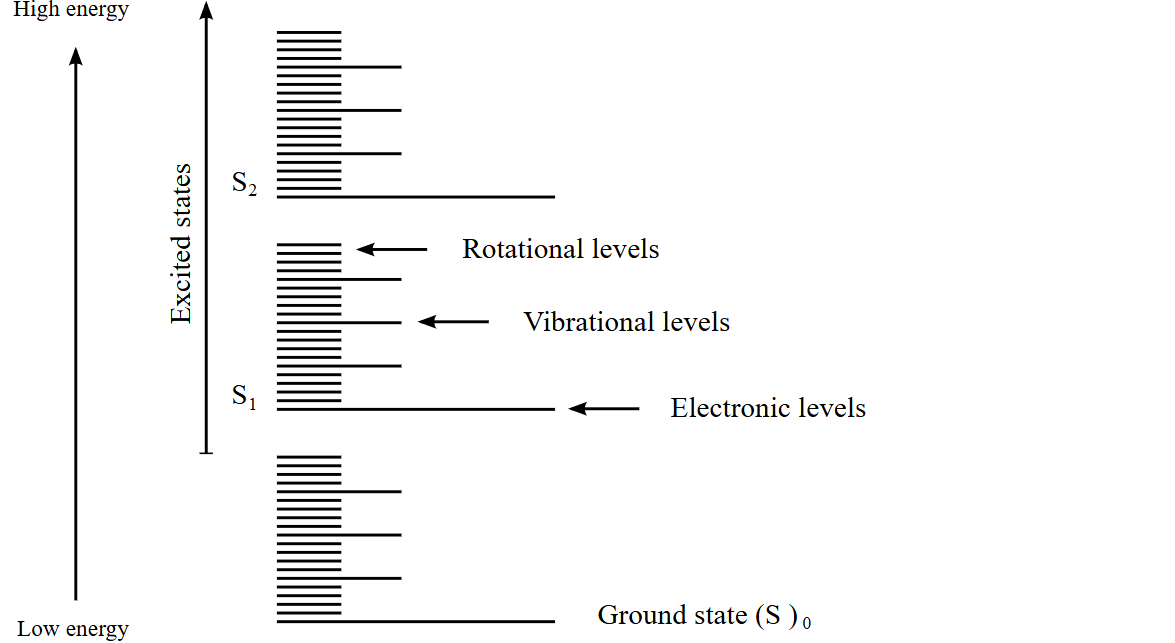 Electrons occupy electronic energy states. Nuclei occupy vibrational or rotational energy states. Electrons nevers occurs in either
vibrational or rotational energy states. Vibrational levels determined via electronic state in the form of:
Electrons occupy electronic energy states. Nuclei occupy vibrational or rotational energy states. Electrons nevers occurs in either
vibrational or rotational energy states. Vibrational levels determined via electronic state in the form of:
|Electronic state, vibrational quantum number>
For example |S0, v=0> or |S1, v=2> etc. Vibrational levels are nuclear motion.For clarification, it is out of our scope but electroncs occurs in vibrational levels as well via coupling. Particularly, electron-nuclear couplings. But nvm for now.
Systems like atoms have only electronic states. No vibrational or rotational states. Diatomic molecules have electronics states + vibrational states + rotational states. Solids (crystals) only have electronic states + vibrational states. Harmonic oscillator models have vibrational + electronics states. And some constrainted systems can be defined with multiple configurations as well.
With these properties, we get to the main idea of optical spectroscopy:
The distances between molecular energy levels are characteristic. They depend on structure of the molecule and interatomic interactions.
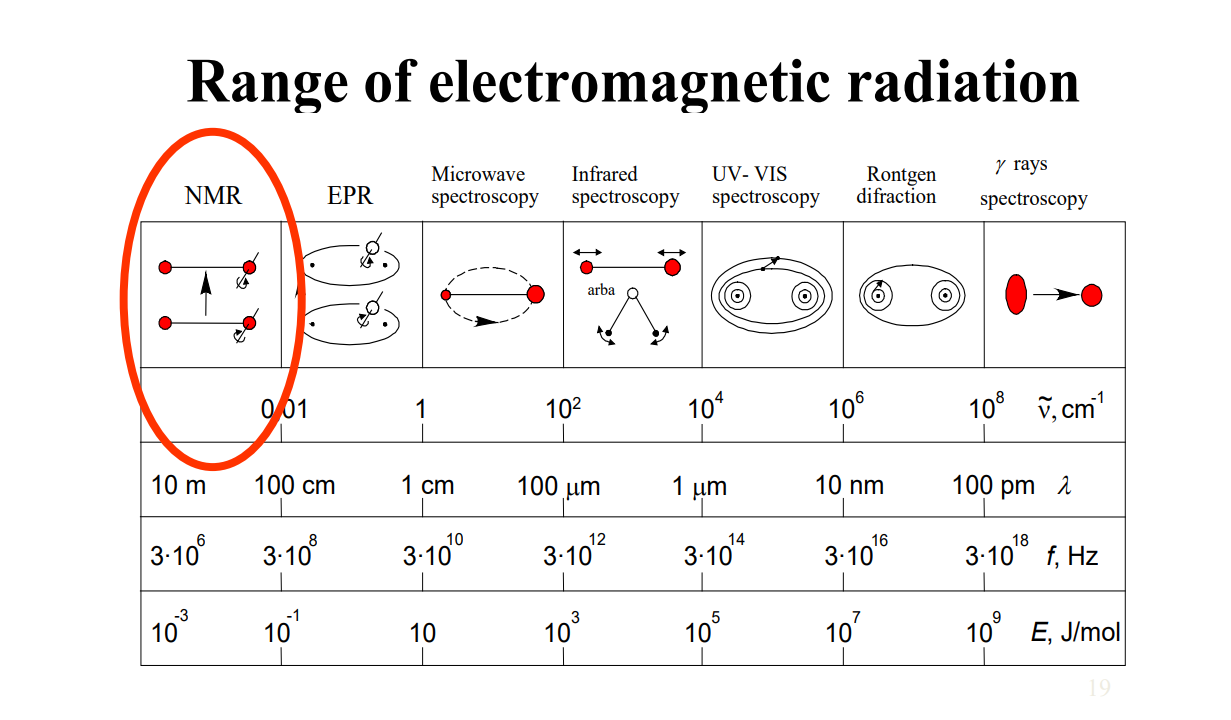
Some analysis for dummies.
We can make some analysis on energy levels in the range. Since we are discussing molecular energy levels for molecules, we are considering all types of transitions as electronics, vibrational and rotational; unlike solids or atoms. So considerably,For NMR range, as energy changes, we expect nuclear spin to flip.
For microwaves, we expect rotational transitions and rotation of molecule.
For IR (infrared), vibrational transitions occurs and bonds stretch. Molecular geometry depended transition. No electron involved.
For Visible and UV ranges, electronic transitions occurs and electroncs are promoted, moved, got excited etc.
For X-ray, I don't even know. But nuclei is tickled.
For Gamma, some nuclear transitions. Nucleus excites.
Test your understanding: Make some analysis in a single region as energy goes up or down, i.e. as frequency increases in visible region what are some highlights in transitions in that region?
So we mostly care about rotations, vibrations and electronic upgrades in molecules. X-rays are interesting as well (X-ray diffraction, X-ray spectroscopy). XRD tells you the positions in a crystal, lattice parameters, molecular structures in solids. X-ray spectroscopy tells you present elements (fingerprints of elements.) There are many techniques. COME BACK HERE IF X-RAY IS IN OUR SCOPE!!!!
Spectroscopy is the main method in materials science.
Since it deals with transitions between vibrational states, it is very much informative. Most informative spectroscopy type is vibrational spectroscopy, indeed.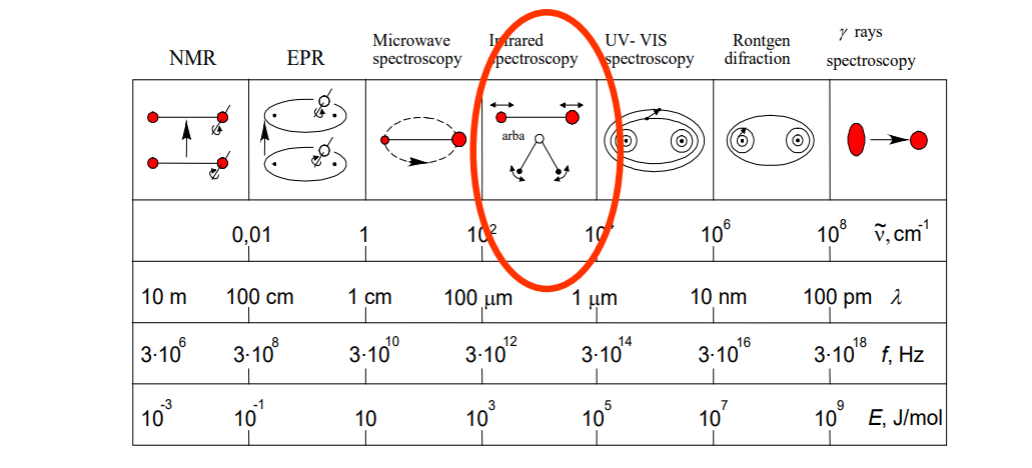
NIR, MIR and FIR
They are classified by their energies scopes, usages and penetration strength. We can continue discussing. Or not.FTIR Spectroscopy
So there is absorption spectroscopy, refering to the method where any substance absorbs any range of electromagnetic radiation. There is also infrared spectroscopy, refering to the range of NIR+MIR+FIR which is around 10000 to 10 cm-1. Now there is also Infrared Absorption Spectroscopy, refering to substances absorbing EM in range of NIR+MIR+FIR.And then we name is synonymously FTIR. End of story.
Vibrational Spectroscopy
Which are IR and Raman Spectroscopy. They are primarly vibrational. But sometimes vibratitional+rotational and even only rotational. But strictly no electronic transitions involved. IR range cannot excite an electron anyways. But Raman, in visible range sometimes possible and we try to avoid it. Avoid how you say? (Check toluene-benzene Raman labwork theory!) We carefully choose our Raman type (either Dispersive Raman or FT-Raman) according to our sample. FT-Raman has weaker signals and uses higher ranges of wavelength (~1064 nm) which avoids exciting electronic states. Dispersive is more free to select any (visible) range (typically lower than 1064 nm) since it has stronger Raman signals and doesn't care about fluoresence.QUESTION: Electronic transition in general is broader than just fluoresence (absorption, emission, decays etc.). Fluoresence is just a type of it. Can you explain how other electronic transitions types are a subject to vibrational spectroscopy (if it is) either in terms of being included or avoided?
#TODO: Determine wavelength vs. wavenumber usage specifically in particular spectroscopies.
#TODO: Analyze some graphs of FTIR, Raman and IR. Add them to the end of the page.
#TODO: Remember the software the prof used that was optimizing molecular structures etc. It was CHEM something.
Introduction to Theoritical Analysis of Molecular Systems
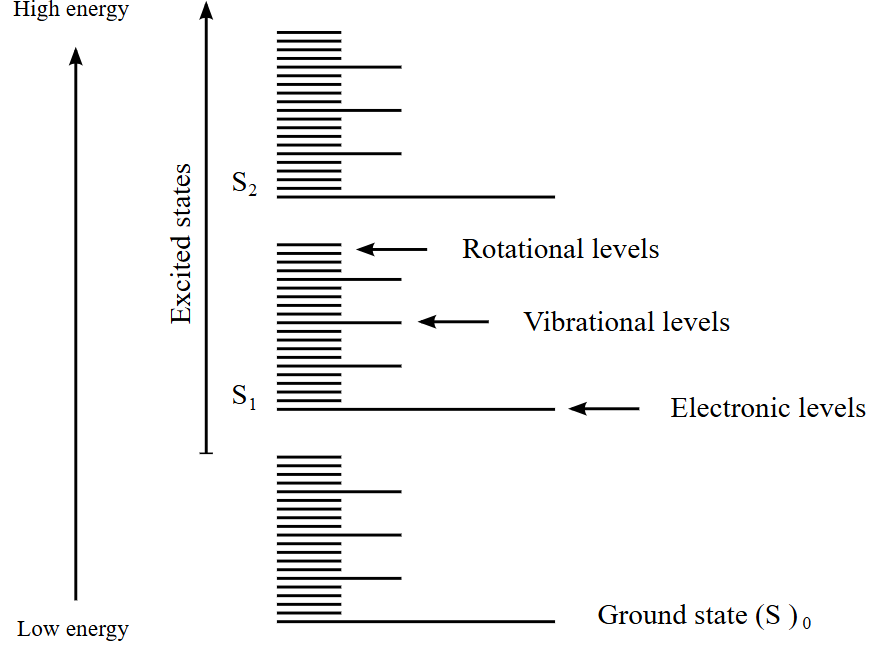
IMPORTANT!!! The main idea in optical spectroscopy is that distance between molecular energy levels depends on the structure of the molecule and intermolecular interactions.
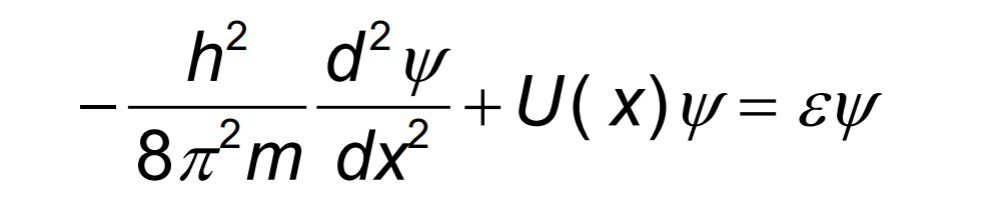 This form of TISE refers to one DOF. If DOF=m>1, we update the Hamiltonian to m-D.
This form of TISE refers to one DOF. If DOF=m>1, we update the Hamiltonian to m-D.
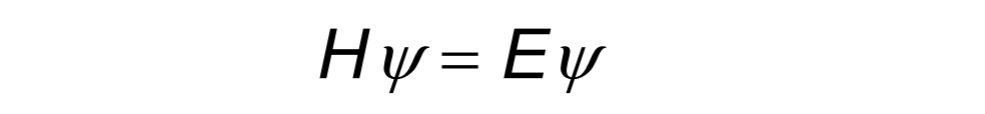 The solution to the Schrödinger equation gives the eigenvalues of energy-certain discreate values of energy; corresponding to energy levels.
The solution to the Schrödinger equation gives the eigenvalues of energy-certain discreate values of energy; corresponding to energy levels.
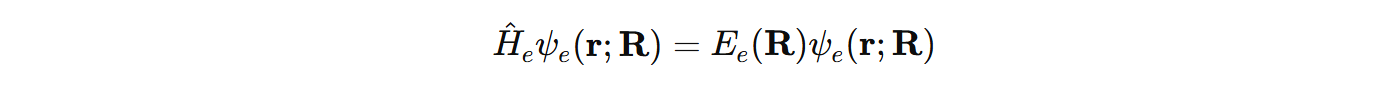 This corresponds to electronic Schrödinger equation. Considerably, all energy levels in discussion are solutions to Schrödinger equation.
This corresponds to electronic Schrödinger equation. Considerably, all energy levels in discussion are solutions to Schrödinger equation.
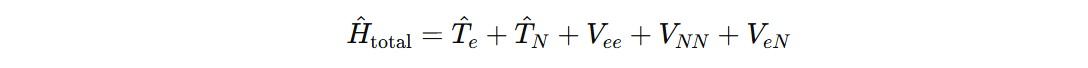 You can think the whole system as the sum for the Hamiltonians. So different Hamiltonians (for example, for vibrational we have harmonic potential and for rotational we have moment of inertia etc.)
You can think the whole system as the sum for the Hamiltonians. So different Hamiltonians (for example, for vibrational we have harmonic potential and for rotational we have moment of inertia etc.)
EXAMPLE
Solve the finite well problem and obtain the eigenvalues and eigenfunctions.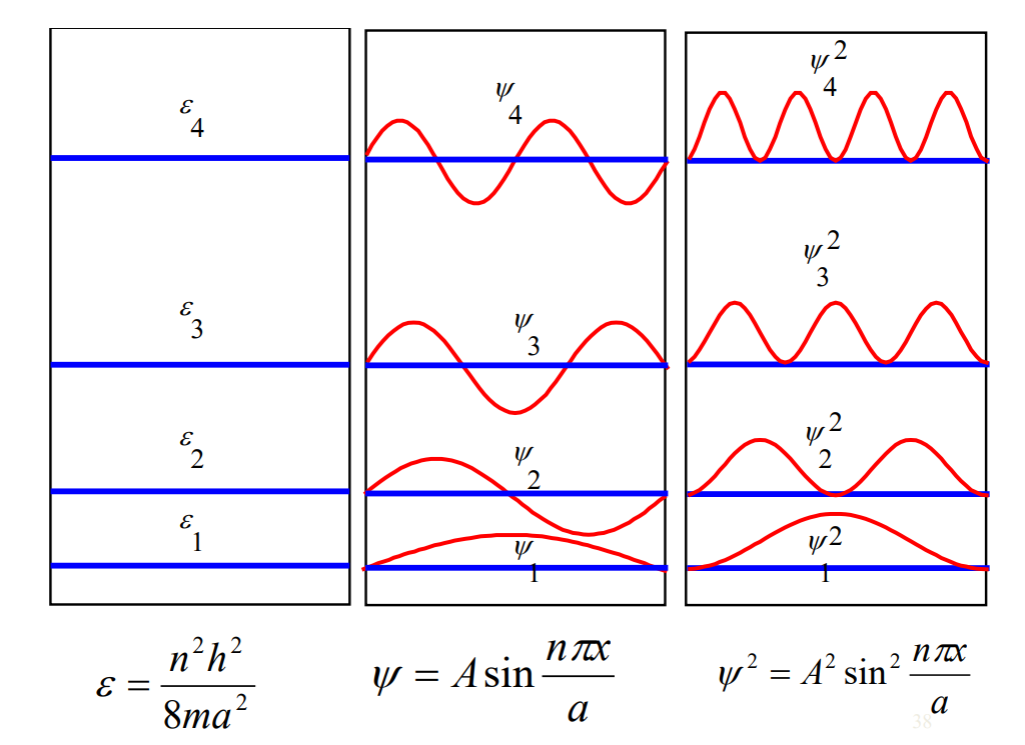
ABSORPTION
Absorption and emission of light by molecule is related to energy levels of the molecule. Einstein proposed a theory explaining this relation.Molecule M absorbs a photon with energy
hf and transiting from energy level m to n; becoming excited molecule M*:
M + hcv -> M*
SPONTANEOUS EMISSION
Excited molecule M* emits photon, transits to lower energy level:M* -> hcv + M
STIMULATED EMISSION
Excited photon emits a photon with INFLUENCE OF ANOTHER EXTERNAL PHOTON.M* + hcv -> M + 2hcv
Now, Einstein Coefficients, are mathematical tools which measure the probability of absorption or emission of light by an atom or molecule.
Einstein coefficients measure the probability of absorption or emission of light by an atom or molecule.
Einstein coefficients measure the probability of absorption or emission of light by an atom or molecule.
STUDY THE MATHEMATICS OF EINSTEIN COEFFCIEINT HERE!!! SLIDE 2 PAGE 41!!!!!
Population of energy states
As system is in thermodynamics equilibrium, it tries to be in the lowest energy level possible -> the ground state.Real molecular systems are not at absolute zero.
But why in real molecular systems the excited energy states are also partly populated? This is called partial population. Which are explain by radiationless transitions. The excess (fazlası) energy is then dissipated (dağılmak) in different forms such as phonons. Which are vibrations associated with lattice vibrations of a solid. Opposite of radiationless transitions are also possible.
BOLTZMANN DISTRIBUTION
Boltzmann distribution explains partial population. Why?The probability of occupying an excited energy level is:
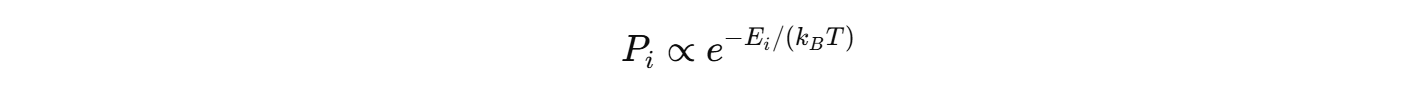 WHY?
A boltzmann distribution is a probability measure that gives the probability that a system will be in a certain state.
WHY?
A boltzmann distribution is a probability measure that gives the probability that a system will be in a certain state.
The ratio of two energy state occupation is expresses as:
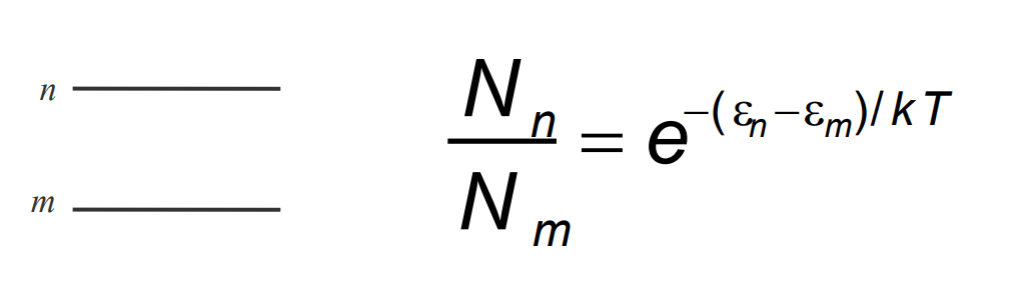
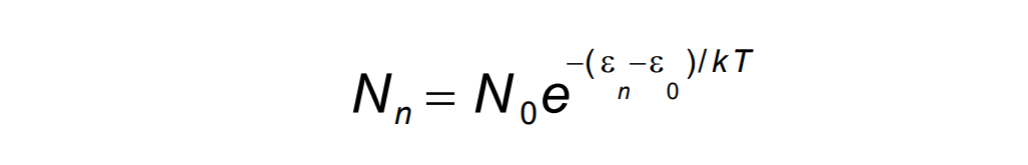 Since some energy levels have same energy (degenerate energy levels) we assign statistical weight to the distribution. Occupation probability (?) of such level becomes:
Since some energy levels have same energy (degenerate energy levels) we assign statistical weight to the distribution. Occupation probability (?) of such level becomes:
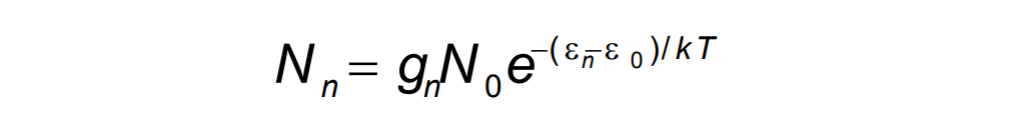 QUESTIONS: Why Boltzmann? What are Nn, Nm? What is T exactly? Temperature of molecule? What is gn? How is it estimated? Is this applicable for any two energy levels? Also how to apply Einstein coefficient here since they are used to measure probability absorption or emission of light. In this Boltzmann distribution, is there a photon?
QUESTIONS: Why Boltzmann? What are Nn, Nm? What is T exactly? Temperature of molecule? What is gn? How is it estimated? Is this applicable for any two energy levels? Also how to apply Einstein coefficient here since they are used to measure probability absorption or emission of light. In this Boltzmann distribution, is there a photon?
SPECTRAL BANDS
What are they?"Intensities of spectral bands are partly determined by population of the initial energy energy level from which the transition starts."
Okay. What the hell is this mean?
"The intensity of an absorption (or emission) spectral line depends on how many molecules occupy the initial energy state from which the transition originates."
This is apperantly the funtamental rule of statistical mechanics (since when?)
Populating or population basically means being occupied by molecules. So as initial state is not populated, no transitions. If it is partially populated, weak transition. And if highly populated, strong transitions, intense peaks. COME BACK HERE MAN... Page 49. Write many questions and analyze please!
FORMULA SHEET